- Home
- About Us
- Work
- Journal
- Blog
- Contact
- Xlstat license key
- Archicad 18 ita
- Metallica discography kickass
- Thebrain 9 development
- Update adobe master collection cs5 windows 7
- Ipadian gamestation pro download
- Gta san andreas extreme edition 2013 free download for pc
- What is fascinating womanhood about
- Myatt app blank
- Odi vilayadu pappa rhymes
- Compuhost v2 upgrade
- Modul bahasa inggris smp kelas 7
- Kodi 17-6 builds 1st generation firestick
- Netcat reverse shell shellshock curl
- Median xl cow level king
- Home
- About Us
- Work
- Journal
- Blog
- Contact
- Xlstat license key
- Archicad 18 ita
- Metallica discography kickass
- Thebrain 9 development
- Update adobe master collection cs5 windows 7
- Ipadian gamestation pro download
- Gta san andreas extreme edition 2013 free download for pc
- What is fascinating womanhood about
- Myatt app blank
- Odi vilayadu pappa rhymes
- Compuhost v2 upgrade
- Modul bahasa inggris smp kelas 7
- Kodi 17-6 builds 1st generation firestick
- Netcat reverse shell shellshock curl
- Median xl cow level king
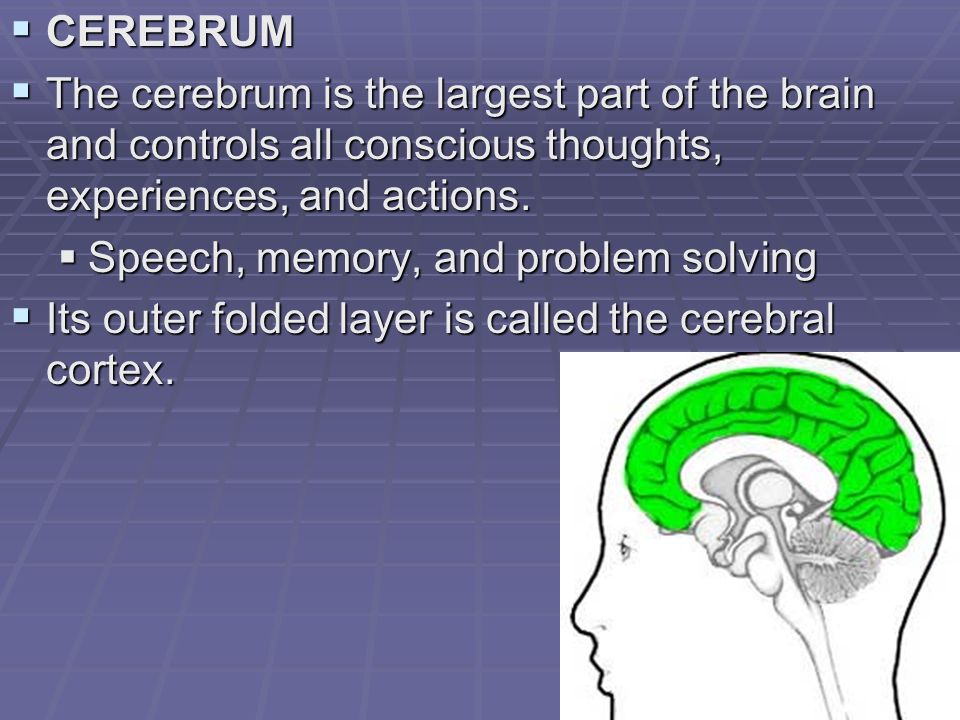
Interruptions to brain blood circulation have been achieved via various techniques including asphyxia, compression of the heart vascular bundle by intrathoracic hook, potassium chloride (KCl) injection and ventricular fibrillation (VF). In order to understand the pathophysiology of brain ischemia and suggest therapeutic approaches, there is a need for an accurate and reliable model of CA in which the variables such as ischemia time and resuscitation environment can be accurately controlled. In those who survive the initial CA nearly 60% die from neurological events and 30% of them suffer significant memory impairment. Stroke and cardiac arrest (CA) are not only the leading causes of death (approximately 800,000 case of stroke and 400,000 out of hospital CA per year in the U.S.), but in the survivors of this ischemic injury, it is linked to an increased risk of cognitive and memory impairment including the long-term effects of dementia. Global Brain Ischemia, Cardiac arrest, Reperfusion, Two phase alternating current, Tau phosphorylation. The findings show the efficiency of this technique in providing a tool to study the further neuronal response to ischemic situations and the underlying mechanisms. We report an early tau dephosphorylation which was followed by hyperphosphorylation after 4 weeks, while the total tau remained unchanged.Ĭonclusions: This method successfully produced reproducible global brain ischemia generating tau dephosphorylation in short-term recovery and hyperphosphorylation in long-term recovery. Results: Resuscitation outcomes were assessed at 1 st and 2 nd hour and 4 th week of restoration of blood circulation and animal survival. Tau and p-tau levels were assessed as an indicator of neuronal response to ischemia.
#THEBRAIN 9 DEVELOPMENT MANUAL#
After 2-, 4- and 8-minute CA, resuscitation was initiated by ventilation, 8 Joules (J) chest electrical shock in the event of VF, adrenaline injection and manual chest compression.ECG, pulse rate and blood oxygen saturation were recorded during the resuscitation. Two phases of AC (24 Volts (V) followed by 18 V) were applied through a two-ring oesophageal wire. This model mimics CA in humans and allows testing of experimental interventions to restore/preserve brain function. Method: In the current study we used a two phase alternating current (AC)-derived reversible CA to generate global-brain ischemia through ventricular fibrillation (VF)/ventricular standstill followed by defibrillation, mechanical ventilation and standard resuscitation in 26 female adult Sprague Dawley rats.

In the current study the effectiveness of a reversible model of CA has been assessed through examining the brain response in expressing tau and hyperphosphorylated tau (p-tau) protein, one of the main hallmarks of Alzheimer's disease.

An accurate, simple and reproducible model of CA ischemia and reperfusion is valuable in assessing the response to ischemia and therapeutic interventions.
Objective: Brain ischemia is a consequence of stroke and cardiac arrest (CA), leading to short and long-term neurological impact involving cognitive function as well as dementia. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

Shohreh Majd, Neuronal injury and repair laboratory, Flinders Medical Centre, Level 4, Rm 4D206, Bedford Park, SA 5042, Australia, E-mail: Int J Neurol Neurother, IJNN-3-040, (Volume 3, Issue 1), Original Article ISSN: 2378-3001 Received: Janu| Accepted: Febru| Published: February 26, 2016Ĭitation: Majd S, Power JH, Koblar SA, Grantham HJM (2016) Introducing a Developed Model of Reversible Cardiac Arrest to Produce Global Brain Ischemia and Its Impact on Microtubule-Associated Protein Tau Phosphorylation at Ser 396.
